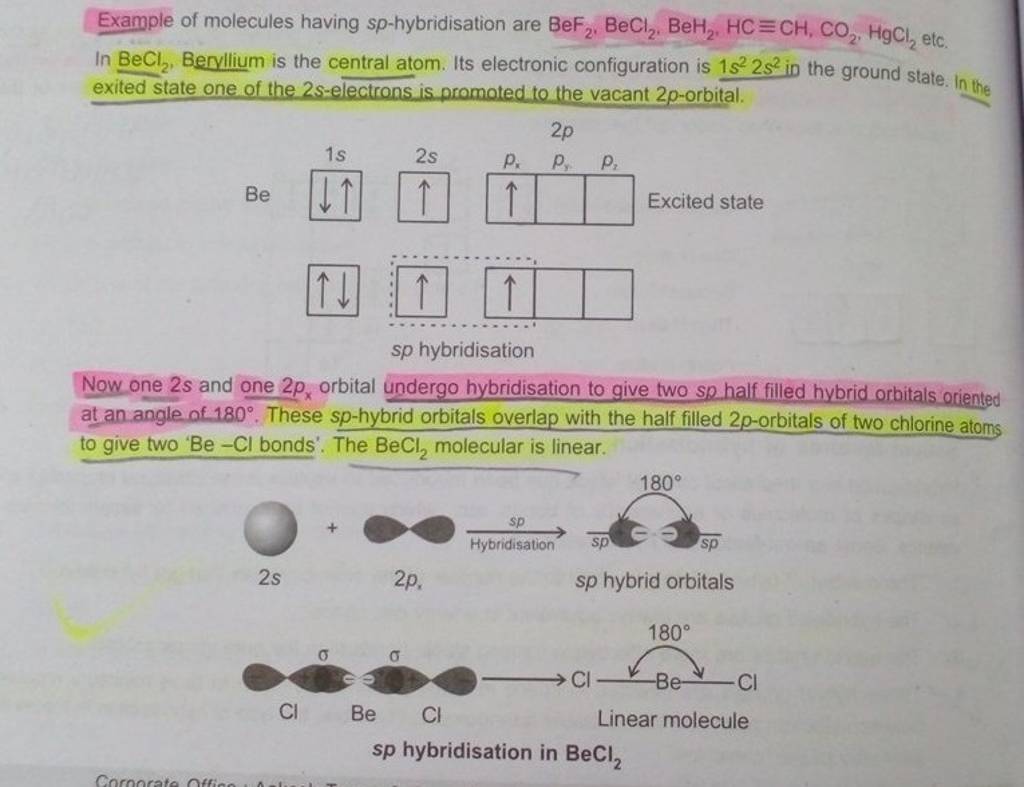
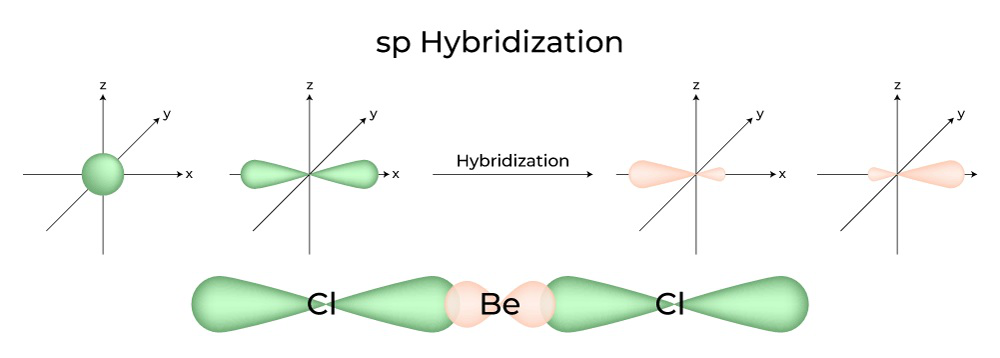
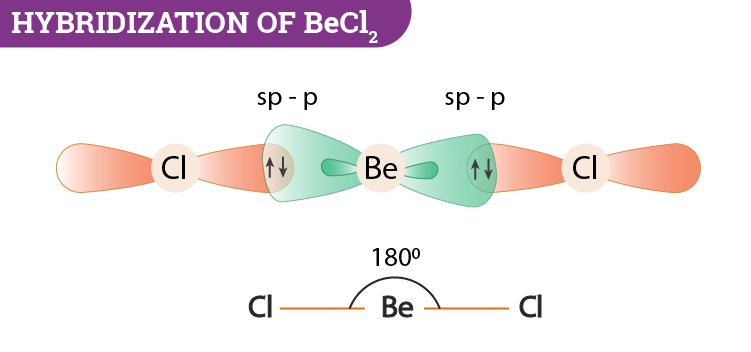
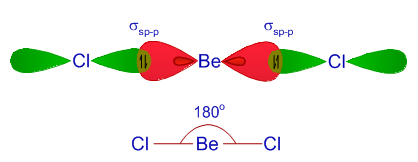
Hybridization Covalent bonds are formed by the sharing of electrons; orbitals overlap to allow for this sharing. The mixing of two or more atomic orbitals. - ppt download

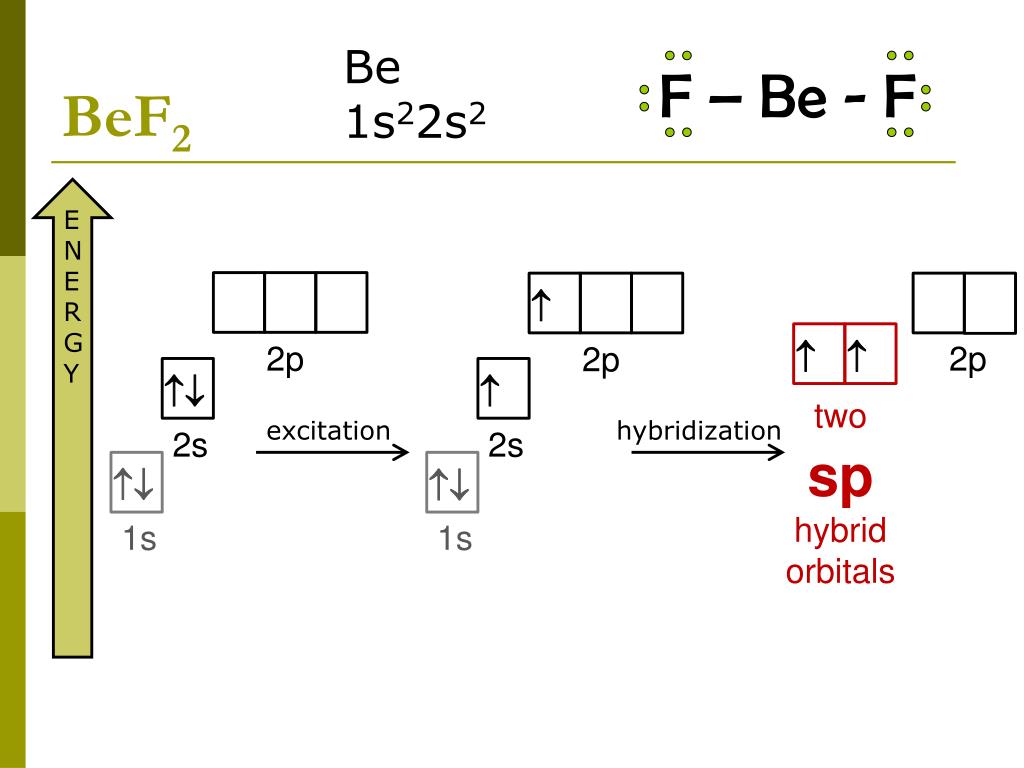
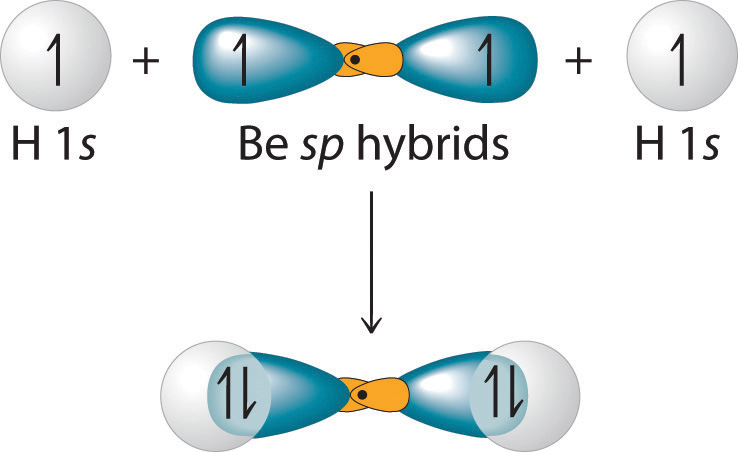
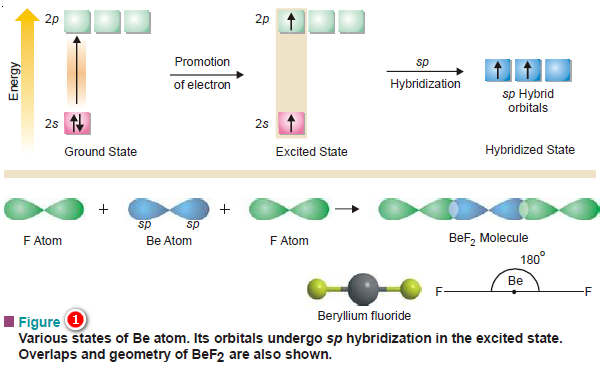
Define Hybridisation State the hybridization the shape of PCl5 and BeF2 - Chemistry - Chemical Bonding and Molecular Structure - 16996831 | Meritnation.com
What is hybridisation ?Explain the formation of BeF2 molecule using hybridisation. - Sarthaks eConnect | Largest Online Education Community