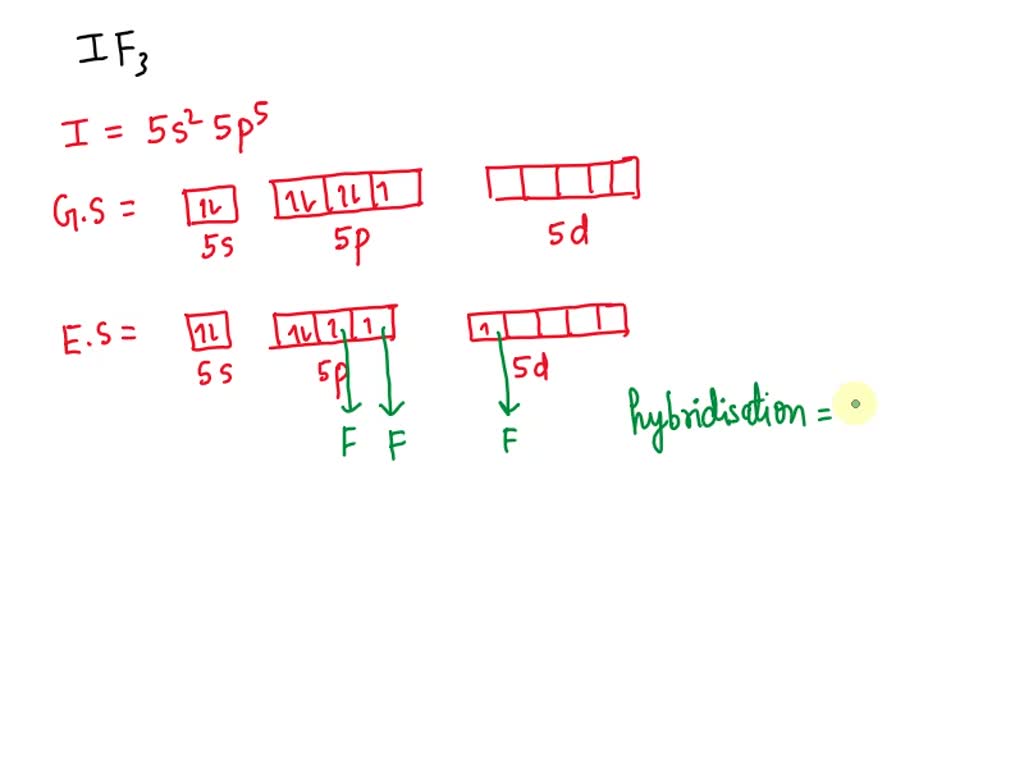
Write down the Lewis structure of ICl3. Provide the electron pair geometry, molecular geometry, and hybridization of iodine. | Homework.Study.com
What is the hybridisation of iodine in IF7 ? Give its structure. - Sarthaks eConnect | Largest Online Education Community
![In the structure of \\[I{{F}_{7}}\\] (Iodine heptafluoride) ----------.(A) \\[{{d}_{xy}},{{d}_{x}},{{d}_{z}}\\] orbitals are involved in hybridization(B) Axial bonds are longer than equatorial bonds(C) There are 10 different orthogonal angels(D)The ... In the structure of \\[I{{F}_{7}}\\] (Iodine heptafluoride) ----------.(A) \\[{{d}_{xy}},{{d}_{x}},{{d}_{z}}\\] orbitals are involved in hybridization(B) Axial bonds are longer than equatorial bonds(C) There are 10 different orthogonal angels(D)The ...](https://www.vedantu.com/question-sets/d7f0d18e-1e05-4073-8dfc-eafee1295a3c8458677129858264325.png)
In the structure of \\[I{{F}_{7}}\\] (Iodine heptafluoride) ----------.(A) \\[{{d}_{xy}},{{d}_{x}},{{d}_{z}}\\] orbitals are involved in hybridization(B) Axial bonds are longer than equatorial bonds(C) There are 10 different orthogonal angels(D)The ...

Hybridization of iodine atoms in ICl_(3) (in its stable form, found in solid state) and I_(2)Cl_... - YouTube
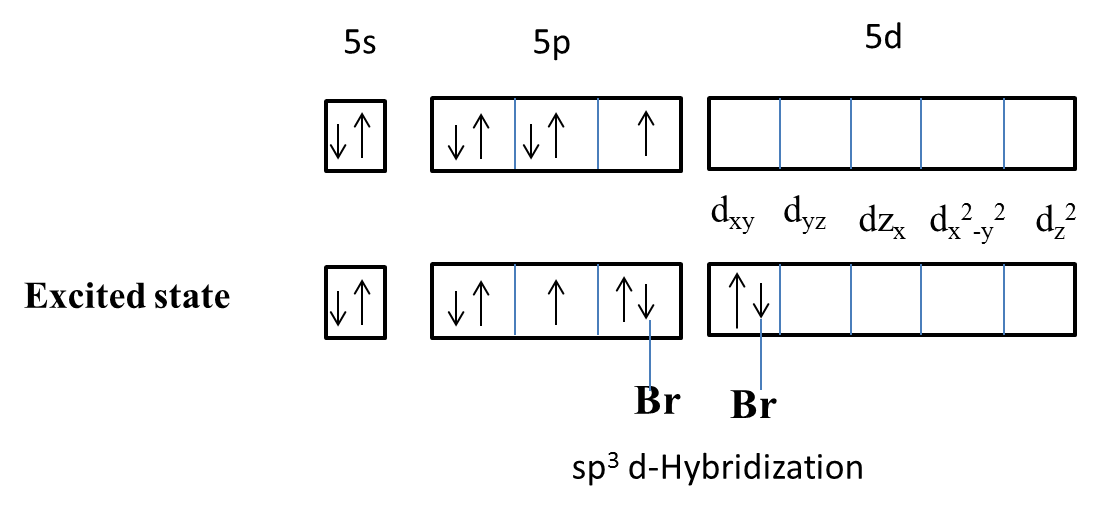
The structure of $IBr_{2}^{-}$ involves hybridization of the type:(A) $s{{p}^{3}}$(B) $s{{p}^{3}}d$(C) $s{{p}^{3}}{{d}^{2}}$(D) None of these

Draw the Structure of If7. Write Its Geometry and the Type of Hybridization. - Chemistry | Shaalaa.com